Fate therapeutics4/27/2023 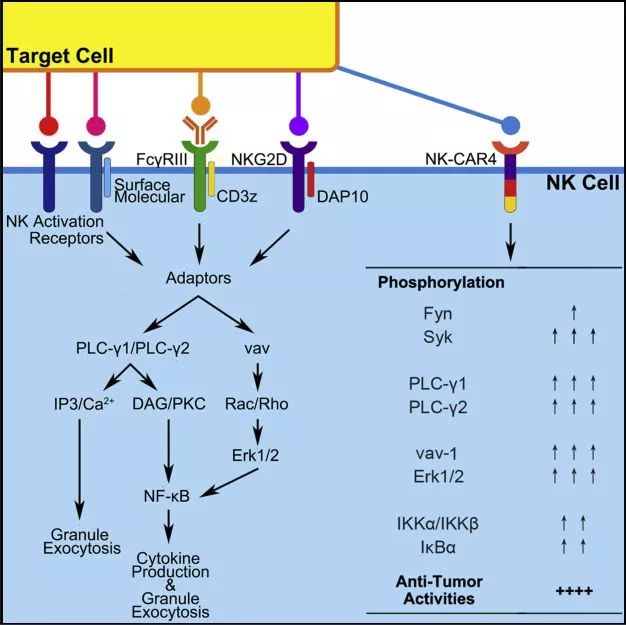 
"We are very excited about the early safety and efficacy data emerging from our groundbreaking clinical study of the first-ever iPSC-derived T-cell therapy. FT819 incorporates several first-of-kind features including the integration of a novel CD19-targeted 1XX chimeric antigen receptor (CAR) construct into the T-cell receptor alpha constant (TRAC) locus, which is intended to promote uniform CAR expression, enhance T-cell potency, and prevent graft-versus-host disease (Eyquem et al. The landmark trial is the first-ever clinical investigation of a T-cell product candidate manufactured from a clonal master induced pluripotent stem cell (iPSC) line, a renewable cell source that enables mass production of engineered T-cell therapies with greater product consistency, off-the-shelf availability, and broader patient accessibility. (NASDAQ: FATE), a clinical-stage biopharmaceutical company dedicated to the development of programmed cellular immunotherapies for patients with cancer, today presented interim clinical data from the dose-escalation stage of its ongoing Phase 1 study of FT819 for patients with relapsed / refractory B-cell lymphoma (r/r BCL) at the 64th American Society of Hematology Annual Meeting and Exposition. 
10, 2022 (GLOBE NEWSWIRE) - Fate Therapeutics, Inc. Interim Phase 1 Dose-escalation Data Indicate Favorable Safety Profile with Complete Responses in Heavily Pre-treated Patients with Aggressive Large B-cell Lymphoma FT819 Off-the-Shelf CAR T-cell Product Candidate Derived from Clonal Engineered Master iPSC Line with Novel CD19-specific 1XX CAR Integrated into TRAC Locus
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
